Botanical Identification
Nymphaea caerulea Savigny — morphological overview and alkaloid localisation
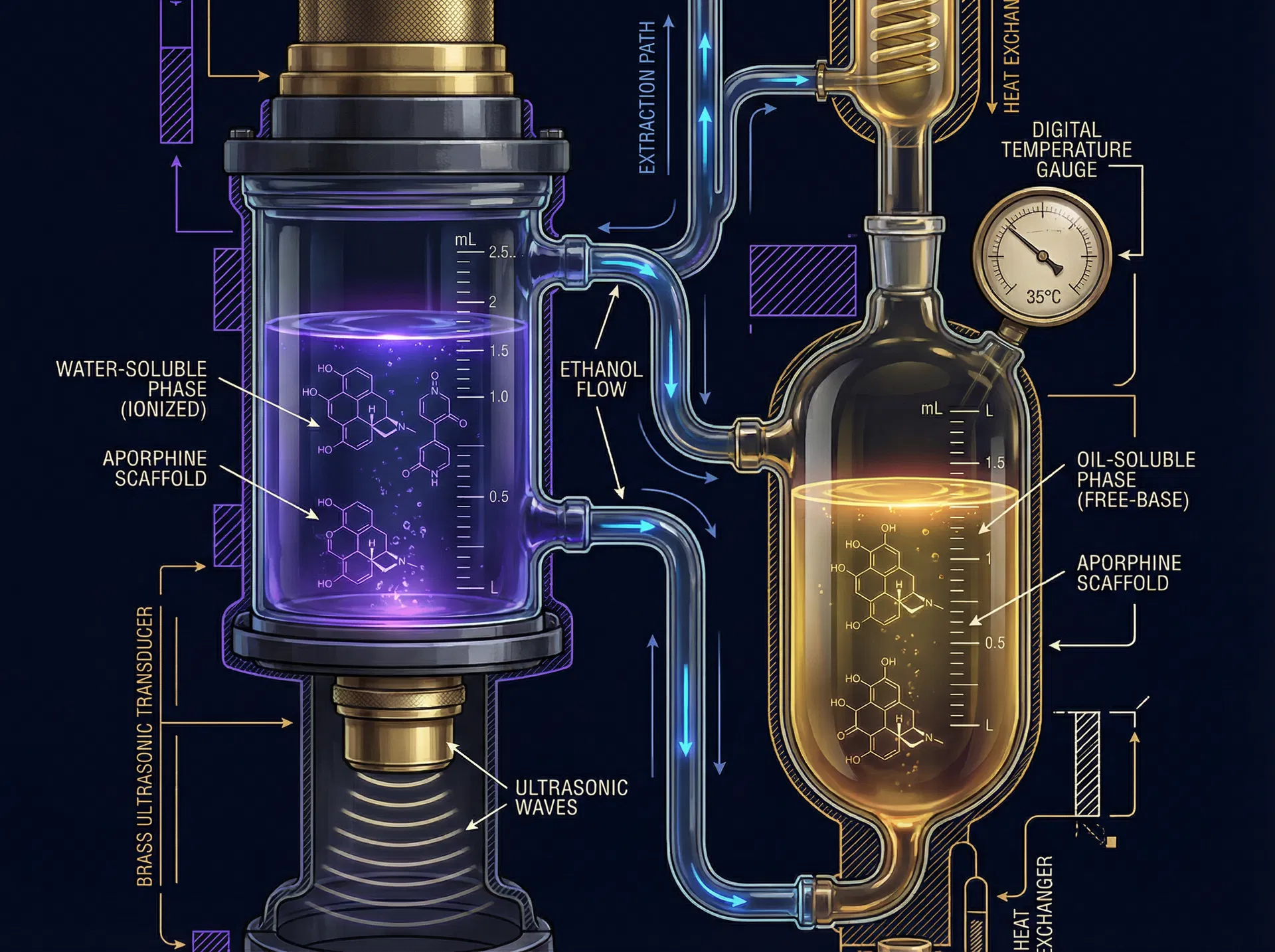
Dual-Phase Extraction Apparatus
95% EtOH · 35°C Ultrasonic Cavitation · 4× Sequential Wash Cycles
Aporphine Alkaloid Structures
Isoquinoline scaffold — shared architecture, divergent receptor selectivity
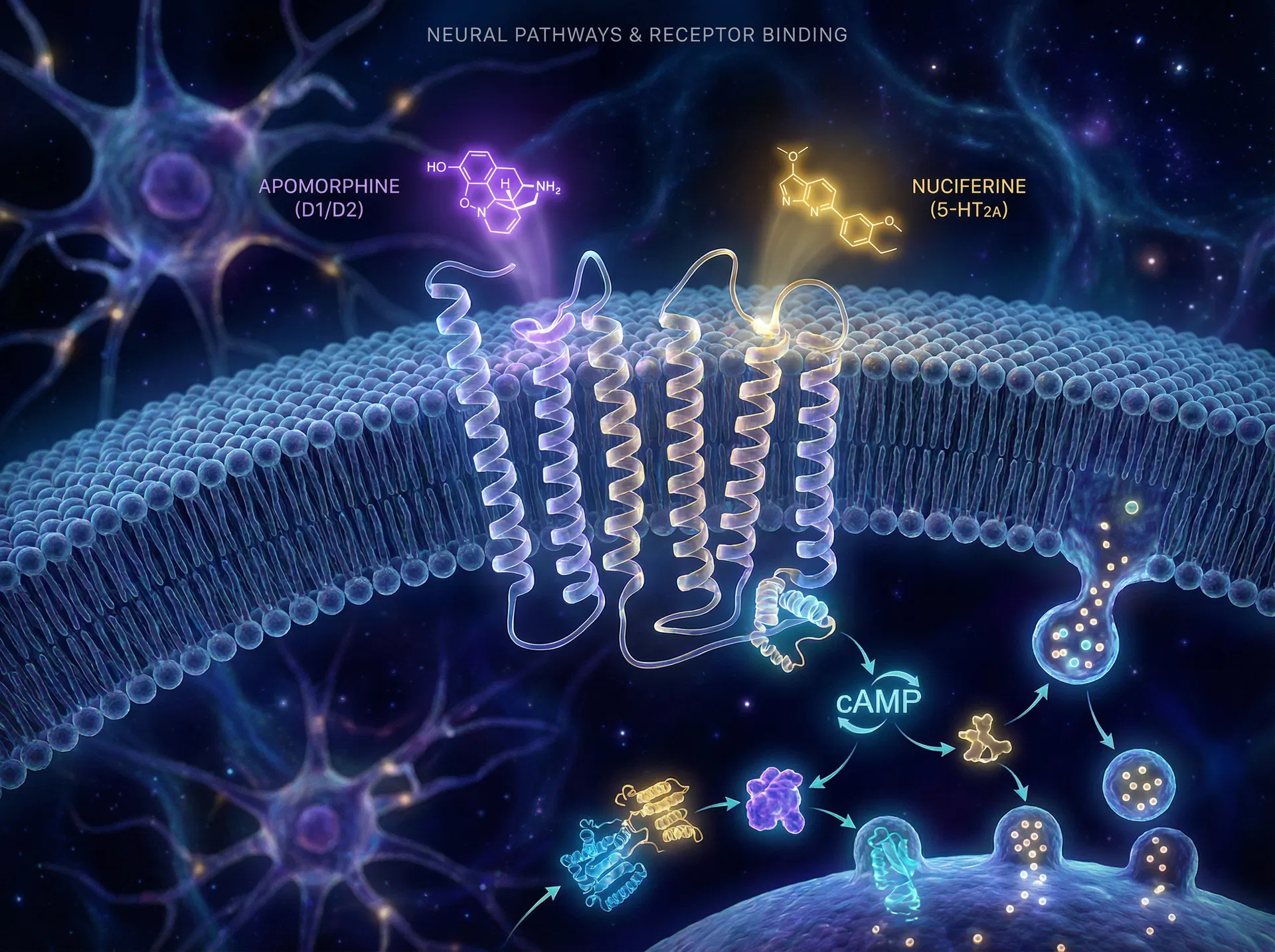
Both alkaloids share the isoquinoline tetracyclic aporphine scaffold. Peripheral substituents — hydroxyl vs. methoxy groups — determine receptor selectivity and polarity class.
Receptor Binding & Neuromodulation
GPCR interaction at dopaminergic and serotonergic receptor sites
Pharmacokinetics & Bioavailability
Route-dependent absorption analysis — oral, sublingual, pulmonary
- Inhalation
- Sublingual
- Oral
Archeo-Technological Correspondence
Ancient Egyptian ritual practice vs. modern pharmaceutical extraction — structural parallels
Experiential State Timeline
Modelled subjective intensity metrics — sublingual / inhalation administration
- Euphoria
- Cognitive Expansion
- Satisfaction
- Integrated Upgrade
System Summary Matrix
Complete pharmacological model — extraction through neuromodulation