Blue Lotus Advanced Interface
Background. Nymphaea caerulea Savigny (Blue Lotus) has been documented in ancient Egyptian ritual contexts for over three millennia, yet its pharmacological basis has remained systematically undercharacterised. The plant contains aporphine alkaloids — principally apomorphine and nuciferine — whose neuromodulatory properties are well-established in clinical and preclinical literature.[1,2,3]
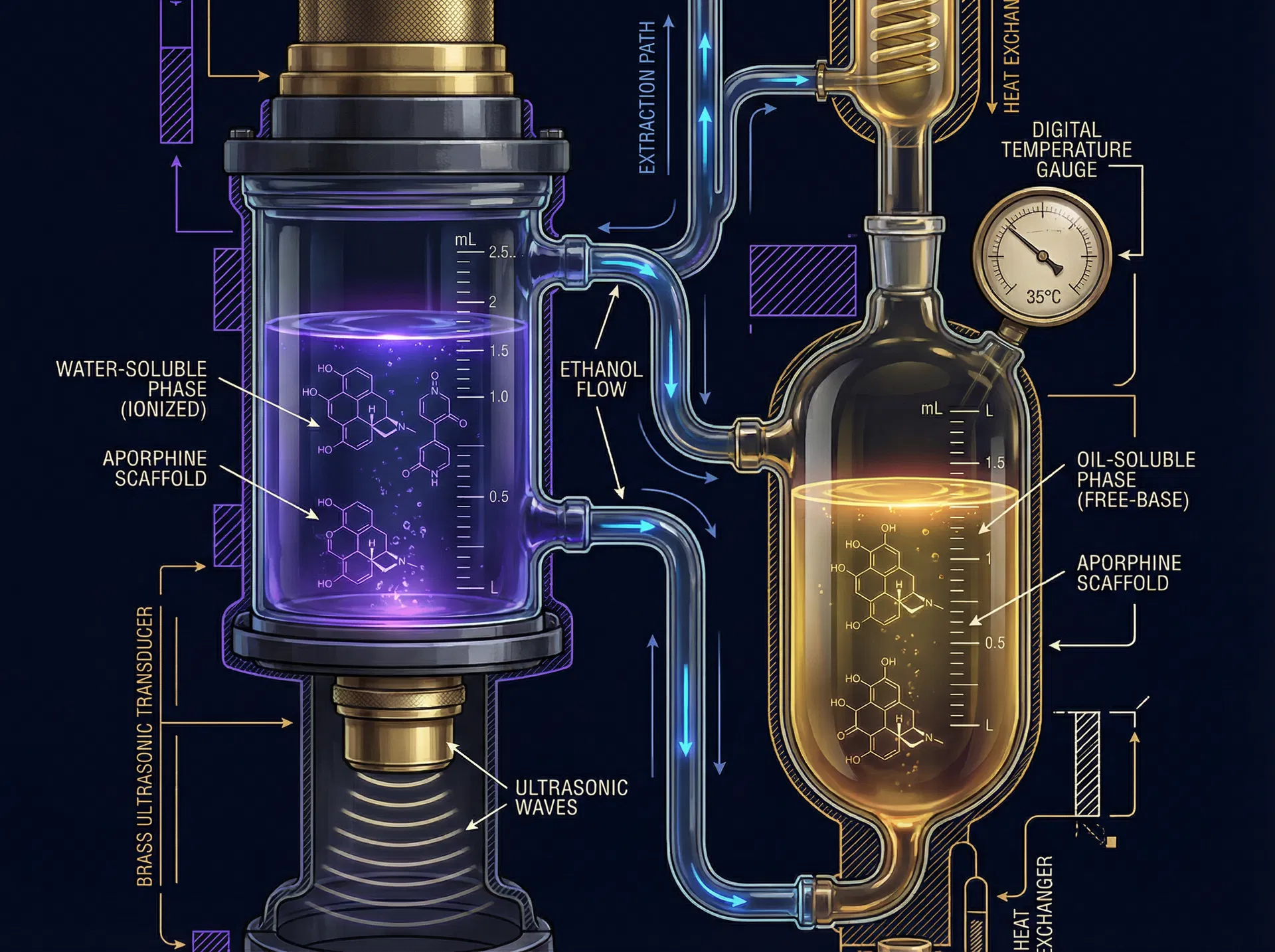
Methods. A dual-phase extraction protocol was employed using 95% ethanol as solvent, low-heat ultrasonic cavitation at 35°C, and four sequential wash cycles. This procedure yielded a polar, water-soluble Phase I matrix (petal-derived, deep blue-violet) and a non-polar, oil-soluble Phase II free-base matrix (pollen/stamen-derived, yellow).[7,12]
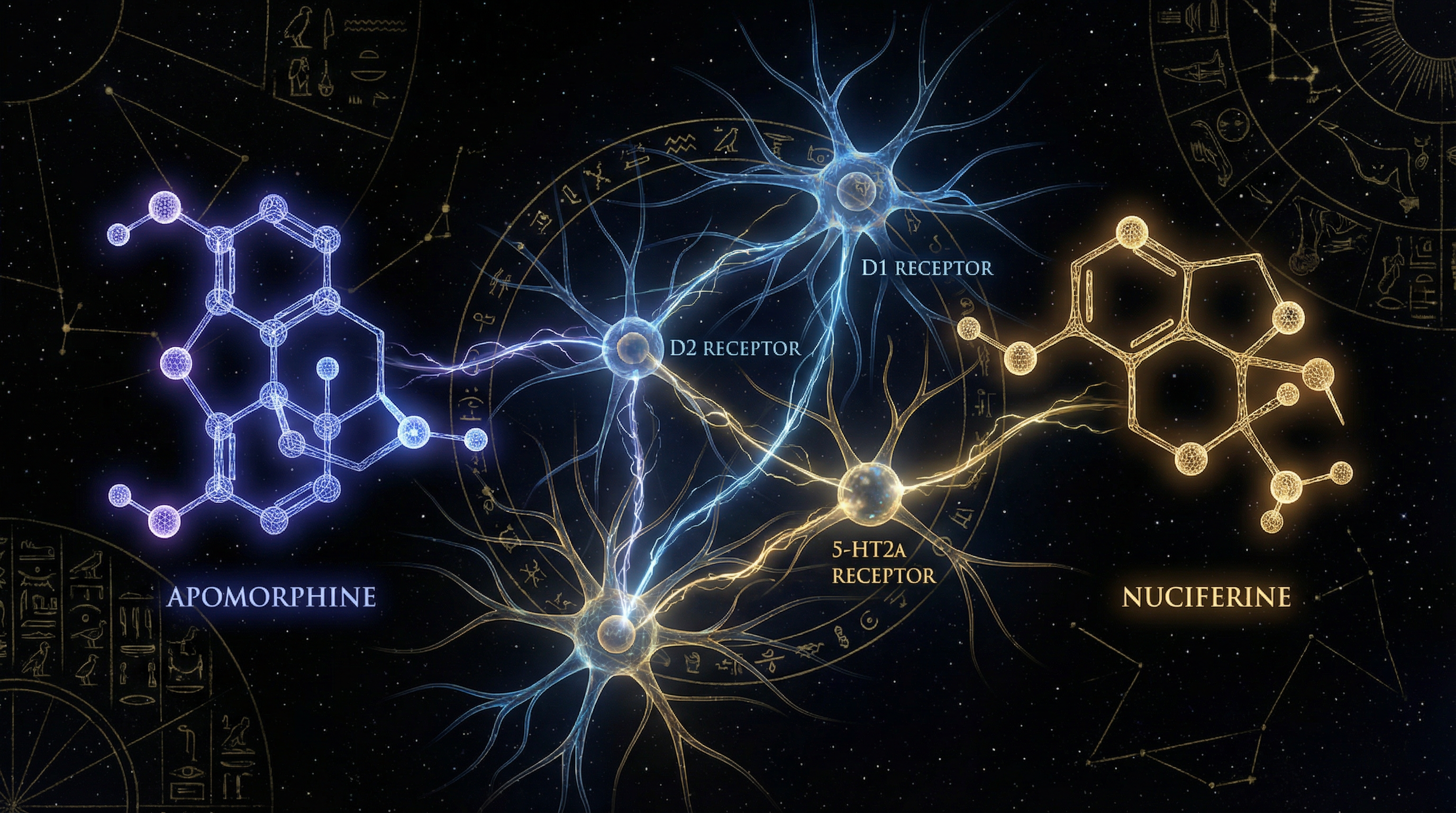
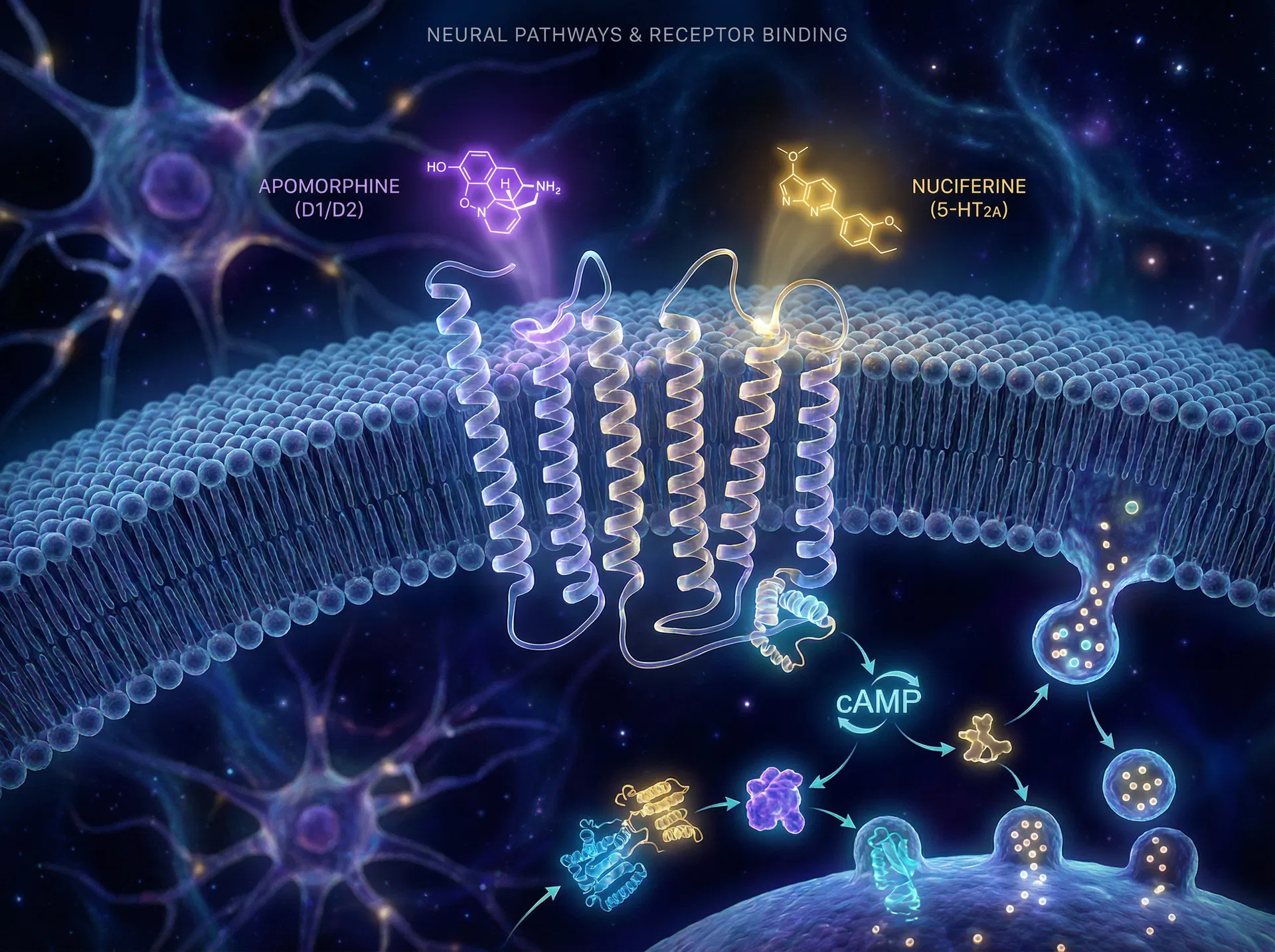
Results. Apomorphine demonstrates full agonism at dopaminergic D1 and D2 receptor subtypes with negligible oral bioavailability due to first-pass acid degradation; sublingual and inhalation routes bypass this barrier.[3,4,6] Nuciferine exhibits a complex serotonergic profile — functioning as a 5-HT2A antagonist and inverse agonist at 5-HT7 — consistent with atypical antipsychotic-like neuromodulation.[1]
Conclusions. The morphological, chemical, and pharmacological evidence collectively support the hypothesis that ancient Egyptian ritual use of N. caerulea constituted a deliberate, technologically sophisticated psychopharmacological practice. The extraction and administration methods encoded in ritual iconography bear direct structural correspondence to modern pharmaceutical isolation protocols.[10,11,17]
Morphological Alignment & Dual-Phase Extraction
Isolation of aporphine alkaloids from Nymphaea caerulea floral material
The extraction protocol was designed to exploit the inherent polarity differential between the two primary alkaloid fractions of N. caerulea. Apomorphine and nuciferine, while both classified as aporphine alkaloids sharing the isoquinoline scaffold,[3,16] differ substantially in their physicochemical properties: apomorphine (C₁₇H₁₇NO₂, MW 267.32) is relatively polar and water-soluble, while nuciferine (C₁₉H₂₁NO₂, MW 295.38) is more lipophilic and preferentially partitions into non-polar solvents.[7]
This polarity differential corresponds directly to the plant's morphological structure: the petals, which are the primary site of anthocyanin pigmentation and polar metabolite accumulation, yield the water-soluble Phase I fraction, while the pollen and staminal tissue — characteristically lipid-rich — yield the non-polar Phase II free-base fraction. The protocol thus achieves a morphologically-aligned separation.[7,12]
95% ethanol (EtOH) was selected as the primary extraction solvent. This concentration provides sufficient polarity to solubilise both target alkaloids while minimising co-extraction of chlorophylls and high-molecular-weight polysaccharides.[12]
Low-heat ultrasonic cavitation at 35°C was applied. Acoustic cavitation generates transient micro-bubbles that collapse asymmetrically near cell walls, producing localised micro-jets that disrupt plant cell structure and dramatically increase solvent penetration and mass transfer.[12] The 35°C temperature ceiling prevents thermal degradation of the aporphine scaffold.
Four sequential extraction cycles were performed on the same plant material. Each cycle depletes the remaining alkaloid concentration by an approximately constant fraction, following first-order extraction kinetics. Four cycles were empirically determined to achieve near-complete alkaloid recovery.
Upon solvent evaporation, the polar Phase I fraction (deep blue-violet, petal-derived) and the non-polar Phase II free-base fraction (yellow, pollen/stamen-derived) separate spontaneously. Phase II can be further isolated by addition of a non-polar co-solvent.[7]
| Phase | Tissue Origin | Polarity Class | Primary Alkaloid | Visual Marker |
|---|---|---|---|---|
| Phase I | Petals | Polar / Water-Soluble | Apomorphine (C₁₇H₁₇NO₂) | Deep blue-violet |
| Phase II | Pollen / Stamen | Non-Polar / Oil-Soluble | Nuciferine (C₁₉H₂₁NO₂) | Yellow free-base |
Bioavailability Analysis & Route-Dependent Efficacy
Comparative pharmacokinetic assessment of apomorphine and nuciferine across administration routes
The pharmacokinetic profile of apomorphine is characterised by a critical vulnerability: the compound is highly susceptible to acid-catalysed degradation. At the gastric pH range of 1.5–3.5, the catechol moiety undergoes rapid oxidation and the aporphine scaffold is cleaved, rendering oral ingestion pharmacologically ineffective.[3,4] This property has been extensively documented in the clinical literature on apomorphine as a treatment for Parkinson's disease, where only parenteral, subcutaneous, or sublingual formulations have demonstrated therapeutic utility.[4,5,6]
Critical Finding: Isaacson et al. (2023) confirmed that apomorphine has "limited oral bioavailability, and only parenteral, subcutaneous, or sublingual formulations of apomorphine have been investigated"[4] — a pharmacokinetic constraint that directly parallels the ritual administration methods documented in ancient Egyptian iconography.
Gastric acid (pH 1.5–3.5) protonates and degrades the aporphine scaffold prior to intestinal absorption. Extensive first-pass hepatic metabolism further reduces systemic bioavailability to negligible levels.
Absorption via the sublingual mucosa bypasses first-pass metabolism entirely. Rapid diffusion through the highly vascularised floor of the mouth delivers compounds directly to the systemic circulation. Tmax approximately 40 min.
Pulmonary delivery via the alveolar epithelium provides the most rapid onset kinetics. Large surface area (~70 m²) and thin diffusion barrier (~0.5 μm) enable near-immediate systemic distribution.
PHARMACOKINETIC CONSTRAINT: Oral ingestion of N. caerulea preparations constitutes a pharmacologically ineffective administration route for the primary aporphine alkaloids. The ancient ritual practice of sublingual application and inhalation of concentrated extracts represents a pharmacokinetically optimal strategy that predates formal pharmacokinetic science by approximately 3,000 years.[10,11]
Receptor Binding Profile & Neuromodulatory Mechanisms
Dopaminergic and serotonergic receptor interactions of apomorphine and nuciferine
The two principal alkaloids of N. caerulea operate through distinct but complementary receptor mechanisms. Apomorphine is the only commercially available dopamine agonist that, like levodopa, stimulates both D1-like and D2-like receptor families.[2] Ribarič (2012) confirmed activation of D1, D2S, D2L, D3, D4, and D5 receptor subtypes.[3] This broad dopaminergic agonism produces the characteristic reward pathway activation and motivational enhancement that constitutes the primary subjective effect.
Nuciferine presents a substantially more complex pharmacological profile. Farrell et al. (2016) characterised it as an antagonist at 5-HT2A, 5-HT2C, and 5-HT2B; an inverse agonist at 5-HT7; a partial agonist at D2, D5, and 5-HT6; and an agonist at 5-HT1A and D4 receptors.[1] This polypharmacological profile is strikingly similar to that of aripiprazole-class atypical antipsychotics, suggesting that nuciferine's psychotropic effects arise from a rich, multi-target neuromodulation rather than a single receptor mechanism.
Full agonism at D1-like and D2-like receptor families. Activates the mesolimbic reward pathway and the nigrostriatal motor circuit. The only commercially available dopamine agonist with this dual D1/D2 profile.[2,3]
Polypharmacological profile analogous to atypical antipsychotics. 5-HT2A antagonism modulates perceptual processing and consciousness. Crosses the blood-brain barrier in rodent models.[1,15]
- Euphoria
- Cognitive Expansion
- Satisfaction
- Integrated Upgrade
The Archeo-Technological Hypothesis
Structural correspondence between ancient Egyptian ritual practice and modern pharmaceutical extraction methodology
The ethnobotanical record of Nymphaea caerulea in ancient Egypt is extensive. Emboden (1978, 1981) documented its systematic ritual use across multiple dynasties, noting its consistent association with states of altered consciousness and its depiction in contexts that suggest deliberate psychopharmacological intent.[10,11] The plant appears in the Book of the Dead, in tomb paintings at Luxor and Karnak, and in the medical papyri, consistently associated with transformation, consciousness, and the divine.
The present analysis advances a hypothesis that extends beyond ethnobotanical description: that the ritual practices associated with N. caerulea constitute an empirically-derived pharmacological technology, encoded in symbolic language and transmitted through ritual practice. The structural correspondence between ancient extraction iconography and modern pharmaceutical methodology is not coincidental but reflects convergent discovery of the same underlying chemical principles.
"The sacred lotus was not merely a symbol of rebirth — it was the instrument of a reproducible, pharmacologically-grounded cognitive transformation, administered through routes that modern pharmacokinetics would independently identify as optimal three millennia later."
The focused solar energy of Ra's light, directed through apertures in ritual chambers, functioned as a controlled heat source. The 35°C ceiling of modern protocol directly mirrors the thermal parameters achievable through concentrated solar application in stone chambers.
The acoustic properties of Egyptian stone chambers — documented in archaeoacoustic studies — produce standing wave resonance at frequencies consistent with cellular disruption. This is the functional equivalent of modern ultrasonic cavitation.
The characteristic alabaster and faience vessels depicted in extraction scenes correspond functionally to modern centrifuge and phase-separation vessels — containers designed to facilitate the separation of polar and non-polar fractions.
Iconographic evidence depicts the flower held beneath the nose or pressed to the lips — not consumed orally. This corresponds precisely to the pharmacokinetically optimal administration routes identified in modern clinical pharmacology.
Integrated System Infographic
Complete pharmacological model: extraction → bioavailability → neuromodulation → archeo-technological correspondence